People with obesity-related heart failure and diabetes can get substantial heart-health benefits from weight-loss drug Wegovy, even if they don’t shed many pounds on the medication, according to new research.
Source link
weight

There may be a stronger case to invest in single stocks over exchange-traded funds in the weight loss space.
Amplify ETFs and Roundhill Investments each filed a prospectus last week to launch funds focused on weight loss companies, a move that Strategas ETF and technical strategist Todd Sohn believes hinges on the performance of two dominant stocks: Novo Nordisk (NVO) and Eli Lilly (LLY).
“The main holdings are going to be Lilly and Novo Nordisk, and probably one or two other big names … along with some of the manufacturers down the supply chain,” he told CNBC’s “ETF Edge” this week. “Ultimately, it’s up to those big behemoths that are playing those drugs.”
With just two players currently at the forefront of the U.S. obesity drug market, ProShares’ Simeon Hyman questions the relevance of weight loss ETFs for investors looking to buy into the industry.
“I think that’s one of the challenges whenever you see an innovation like this,” the firm’s global investment strategist said in the same interview. “If the benefits are going to incumbents, then maybe there isn’t a theme per se that needs to be exploited.”
Strategas’ Sohn also suggested that ETFs based on themes, rather than sectors or indices, might be falling out of favor with investors.
“I think thematics are a little bit on the backburner right now, especially the way they performed the last couple of years. I think there’s room for them, but more than one, it’s gonna be tough,” he said.
So far in 2024, Novo Nordisk has gained 29% and Eli Lilly is up 30%, as of Wednesday’s close. The broader Health Care Select Sector SPDR (XLV) is 7% higher during the same period.
Disclaimer
Patients Regain Weight After They Quit Using Ozempic Rival Zepbound. Here’s Why That Makes Eli Lilly Stock an Even Better Buy.
Seemingly bad news can sometimes be really good news. I think a recent study for Eli Lilly‘s (LLY -0.28%) rival to Novo Nordisk‘s massively successful Ozempic and Wegovy provides a great case in point.
Results from the Surmount-4 clinical trial were published on the Journal of the American Medical Association‘s website on Dec. 11, 2023. Researchers found that patients who took Lilly’s weight loss drug tirzepatide (marketed as Zepbound for weight loss and Mounjaro for type 2 diabetes) regained roughly half of their weight after they stopped taking the drug for one year.
Investors reacted negatively to the news. Lilly’s share price fell around 5% before rebounding somewhat. However, my view is that there’s more to the story that many investors are missing. Here’s why the recent data actually makes Lilly stock an even better buy.
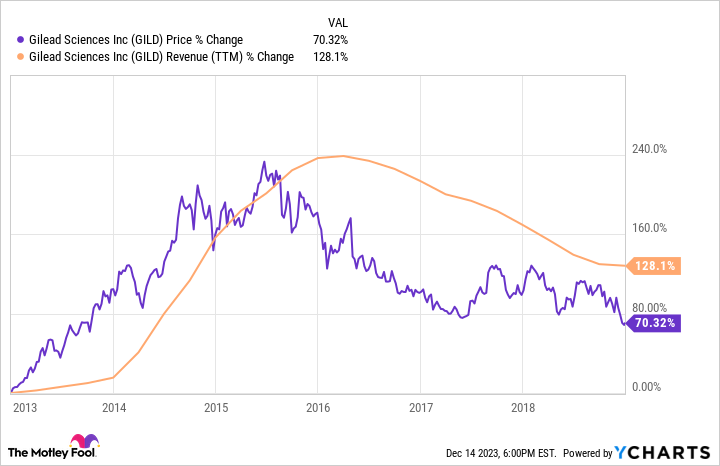
No Gilead effect here
To make my argument, let me first point to a completely opposite scenario that I think is instructive. A decade ago, Gilead Sciences (GILD -1.77%) won U.S. Food and Drug Administration (FDA) approval for Sovaldi. It was the first therapy that effectively cured hepatitis C for many patients.
Over the next few years, Gilead launched even more powerful combination therapies for treating hepatitis C virus (HCV) infection. Harvoni, Epclusa, and Biktarvy — along with Sovaldi — all became blockbusters. Gilead raked in money hand over fist.
Everything went great for Gilead Sciences for a while. But then, the company’s sales began to decline, as did the price of the biotech stock. What happened?
A person who is cured of hep C doesn’t need medications anymore. Gilead began to run out of patients for its HCV drugs.
As I mentioned, this is the opposite scenario than we’re seeing for Eli Lilly and Zepbound. Patients have ample motivation to continue taking Lilly’s drug. If they don’t, they’re likely to regain their lost weight.
This means that Lilly isn’t going to experience the “Gilead effect.” Sales for Zepbound should remain strong as patients continue taking the drug to keep their weight down.
No one should have expected a different outcome, though. Research has also shown that patients who stop taking Novo Nordisk’s Ozempic and Wegovy regain weight. In addition, Lilly Chief Scientific and Medical Officer Dan Skovronsky stated earlier this year that “unfortunately, tirzepatide is probably like every other drug we have which requires you to take it to continue to get the benefits.” His use of the word “unfortunately” applies to patients and payers — it’s very fortunate for Lilly and its shareholders.
Lilly’s abundance of riches
The combination of Mounjaro and Zepbound could generate peak annual sales of more than $50 billion if analysts’ projections are on target. Lilly could very well soon have the world’s best-selling drug.
But while Mounjaro/Zepbound is the company’s crown jewel, Lilly has an abundance of riches in its current product lineup and pipeline. Verzenio is already a blockbuster in treating breast cancer, with sales skyrocketing 68% year over year in the third quarter of 2023. It’s in late-stage testing for treating prostate cancer, as well.
Sales for two other cancer therapies — Retevmo and Tyvyt — soared at least 50% year over year in Q3. Lilly’s type 2 diabetes drug Jardiance continues to perform well, with sales jumping 22% in the recent quarter. The company remains a major player in the autoimmune disease market with Taltz and Olumiant enjoying solid momentum.
The big drugmaker hopes to soon win FDA approval for donanemab in treating early stage Alzheimer’s disease. It seeks to add another autoimmune disease drug to its portfolio with lebrikizumab in treating atopic dermatitis.
Lilly could further boost its type 2 diabetes and weight loss franchise with orforglipron, an oral therapy that’s in phase 3 clinical studies. And those are just a few of the company’s promising pipeline candidates.
That seemingly bad news with the data released recently about tirzepatide truly is good news for Eli Lilly. I expect that the drugmaker and its shareholders will have plenty of more good news on the way over the next few years.
Keith Speights has no position in any of the stocks mentioned. The Motley Fool has positions in and recommends Gilead Sciences. The Motley Fool recommends Novo Nordisk. The Motley Fool has a disclosure policy.
Eli Lilly (LLY 0.12%) and Novo Nordisk (NVO -0.46%) have been the two best-performing large-cap pharma stocks over the past 10 years. The core reason is their strong competitive positions in type 2 diabetes and obesity care.
Lilly’s leading metabolic disease therapy is tirzepatide, which is sold under the brand names Mounjaro for type 2 diabetes and Zepbound for obesity care. Bullish analysts expect this lead metabolic therapy, combined with Lilly’s follow-on offerings in the space, to hit $65 billion in annual sales by 2031.

Image source: Getty Images.
Novo’s main product in this segment is semaglutide, which is available as Wegovy for weight loss, Ozempic as a once-weekly injection for type 2 diabetes, and Rybelsus as a once-daily tablet for type 2 diabetes.
Although Novo holds a first-mover advantage for this class of drugs in both the weight loss and type 2 diabetes settings, the Danish drugmaker is expected to essentially split these markets with Lilly by 2031.
Which of these supercharged growth stocks is the better investment option right now? Let’s examine them more closely to find out.
A traditional and nontraditional valuation comparison
Even though Novo and Lilly both operate in the pharmaceutical industry, these two healthcare titans have dramatically different approaches to value creation. Novo focuses primarily on diabetes and obesity, while Lilly has a more diversified portfolio of drugs. How do they compare in terms of valuation and innovation?
One way to compare them is to use a three-factor model, which estimates the future earnings of a company based on its growth rate, costs, and shareholder rewards. Using this method, Novo’s stock seems to be cheaper than Lilly’s, as it trades at a lower range of multiples for its projected 2031 earnings (midpoint estimate of 16.5) compared to those for Lilly’s stock (midpoint estimate of 28). However, this is a rough approximation that depends on several assumptions that may fail to pan out.
Another way to compare them is to look at their research and development (R&D) spending, which partially reflects their investment in innovation. Innovation is crucial for pharma companies, as their products have limited patent protection. What’s more, R&D spending can be seen as a simple, yet useful, proxy for the net present value (NPV) of a company’s pipeline, which is the sum of the projected cash flows from its clinical portfolio on a risk-adjusted basis.
Lilly has been investing heavily in R&D, spending about a quarter of its annual sales on developing new drugs over the past five years. Novo, on the other hand, has been more conservative, spending only about 13% of its sales on R&D over the same period.
This difference reflects the broader scope of Lilly’s clinical efforts relative to Novo’s over this period. Apart from diabetes and weight loss, Lilly has also been investing in multiple high-value areas such as cardiovascular care, immunology, neuroscience, and oncology. Novo, on the other hand, has been mainly focused on building on its dominance in type 2 diabetes treatment and gradually branching out into therapies for rare diseases.
Verdict
Lilly’s higher premium is arguably well deserved. The company’s substantially larger R&D spend as a fraction of total revenue, and resulting broader pipeline, make it less dependent on the future — and largely unknown — competitive dynamics of the type 2 diabetes and/or weight loss markets. So while Novo’s more specialized approach to pipeline development should translate into a solid competitive position in these high-profile disease settings, Lilly’s stock is arguably the better buy because of the company’s broader and less risky approach to growth.
Eli Lilly’s drug tirzepatide was approved for weight loss by the FDA.
Courtesy of Eli Lilly
Patients who took Eli Lilly‘s weight loss drug Zepbound regained around half the weight they shed after stopping the newly approved treatment for a year, according to data released Monday.
The data, which represents the full results from an 88-week study funded by Eli Lilly, suggests that people have to stay on the weekly injection if they want to maintain significant weight loss.
Shares of Eli Lilly closed more than 2% lower Monday after the data was published in the research journal JAMA. The pharmaceutical giant released initial results from the same study, which was conducted by some Eli Lilly employees and some outside researchers, in July.
Zepbound, Novo Nordisk‘s weight loss injection Wegovy and their blockbuster diabetes counterparts have soared in popularity, and in turn have run in short supply in the U.S. over the past year because they help patients lose substantial weight without surgery. Some Wall Street analysts believe Zepbound, which uses the same active ingredient as Eli Lilly’s diabetes drug Mounjaro, could become the best-selling drug of all time.
People who discontinue Wegovy and Novo Nordisk’s diabetes medication Ozempic have also regained weight, raising concerns among U.S. health insurers about the high costs involved with long-term coverage of the pricey drugs.
The study on Eli Lilly’s treatment showed that 670 obese patients without diabetes lost around 20% of their body weight on average after taking Zepbound for 36 weeks. Half of those patients then continued the drug for another 52 weeks, while the other half switched to a placebo for the next year.
Patients who continued Zepbound lost an additional 6.7% of their weight on average from weeks 36 to 88, while those who stopped taking the drug regained 14.8% of their weight.
Still, those who discontinued Zepbound still ended the 88-week study with 9.9% less weight than they started with, indicating that they only regained about half the weight they initially lost.
“If you look at the magnitude of the weight gain, they gain back about half the weight they had originally lost over a one-year period of time,” lead study author Dr. Louis Aronne, an obesity medicine specialist and professor of metabolic research at Weill Cornell Medicine in New York City, said in an interview with CNN.
About 17% of those who stopped Zepbound maintained at least 80% of their original weight loss, the study said. Meanwhile, 9 in 10 of the people who continued Zepbound were able to maintain at least 80% of the weight they lost.
Throughout the full 88-week study, health-care professionals encouraged all patients to cut about 500 calories per day from their diet and exercise at least 150 minutes a week.
“Patients, providers and the public do not always understand obesity is a chronic disease that often requires ongoing treatment, which can mean that treatment is stopped once weight goals are met,” said Dr. Jeff Emmick, senior vice president of product development at Eli Lilly, in a statement.
But Emmick said Monday’s study shows that “continued therapy can help people living with obesity maintain their weight loss.”
Don’t miss these stories from CNBC PRO:
Sopa Images | Lightrocket | Getty Images
Pfizer’s twice-daily version of its experimental weight loss pill has now joined a long list of other scrapped drugs that aimed to treat obesity but came with unintended consequences.
The drugmaker on Friday said it will stop developing the twice-daily treatment, danuglipron, after obese patients taking the drug lost significant weight but experienced high rates of adverse side effects in a midstage clinical trial. Pfizer noted that it will release data on a once-daily version of the pill next year, which will “inform the path forward.”
The announcement came six months after Pfizer scrapped a different once-daily pill in June, citing elevated liver enzymes. Pfizer’s move to drop two obesity drug candidates in just a few months demonstrates how difficult it is to develop an effective, safe and tolerable treatment for losing weight, even after recent breakthrough medications entered the space.
That includes Novo Nordisk‘s Wegovy and diabetes treatment Ozempic as well as Eli Lilly‘s diabetes drug Mounjaro. They have all skyrocketed in popularity — and slipped into shortages — over the last year for safely and successfully causing significant weight loss. An estimated 40% of U.S. adults are obese, making those drugs the pharmaceutical industry’s newest cash cow.
But before the current weight loss industry gold rush, the path to treating obesity was strewn with failures dating back decades.
The main reason many experimental treatments were scrapped by drugmakers, rejected by U.S. regulators or eventually pulled from the market were unintended side effects, including elevated liver enzymes, cancer risks, cardiovascular risks and serious psychiatric problems, such as suicide.
Eisai’s lorcaserin
One of the most recent casualties among experimental obesity drugs is Japanese drugmaker Eisai’s lorcaserin, which was removed from the market in 2020 due to causing an increased risk of cancer in patients.
The Food and Drug Administration greenlit lorcaserin in 2012 based on several clinical trials but required Eisai to conduct a larger and longer study on the drug after the approval.
That study on about 12,000 patients over five years found that more people taking lorcaserin were diagnosed with cancer compared with those taking a placebo, which led the FDA to pull the drug from the market.
Lorcaserin, marketed under the brand name Belviq, didn’t appear to gain much traction while it was commercially available. In its full-year 2019 earnings, Eisai reported that Lorcaserin had sales of $28.1 million in the U.S. for the year. Global sales of the drug were about $42 million. Eisai’s total sales for the year were roughly $4.42 billion.
Sanofi’s rimonabant
An obesity drug called rimonabant from Sanofi and Aventis was withdrawn from all markets in 2008 due to the risk of serious psychiatric problems, including suicide.
Notably, the treatment never won approval in the U.S. because a panel of experts to the FDA rejected the drug amid fears that it may cause suicidal thoughts. But European regulators approved rimonabant, marketed under the name Acomplia, in 2006 based on extensive clinical trials.
Two years later, European regulators recommended the suspension of rimonabant after one of its committees determined that the risks of the treatment — particularly psychiatric issues — outweighed its benefits.
The treatment suppressed appetite by blocking the receptor of cannabinoid substances in the brain, which plays an important role in regulating the body’s food intake and metabolism.
Due to rimonabant’s limited time on the market and failure to win U.S. approval, the drug never reached Sanofi’s lofty projection that it would eventually generate $3 billion a year or more.
Abbott Laboratories’ sibutramine
Several obesity drugs have also been discontinued, rejected or pulled from the market due to unintended cardiovascular risks.
That includes sibutramine from Abbott Laboratories, which was once widely used as a treatment for obesity along with diet and exercise.
The drug was first approved in 1997, but carried warnings about high blood pressure and a risk of heart attack and stroke in cardiovascular patients.
A large, long-term trial on nearly 10,000 adults confirmed that sibutramine was associated with a significant increase in cardiovascular events, which prompted regulators in the U.S. and Europe to pull the drug from those markets in 2010.
Sales of sibutramine had been dwindling ahead of its removal from the market. The drug raked in only $80 million globally, including $20 million from the U.S., in the first nine months of 2010.
Recent evidence suggests that the newest slate of approved weight loss drugs may have the opposite effect on heart health: Weekly injections of Wegovy slashed the overall risk of heart attack, stroke and death from cardiovascular causes by 20%, according to a recent clinical trial.
Weight loss has always been big business, but it’s exploded of late due to surging demand for Ozempic, Wegovy and other new diabetes and obesity drugs.
In the first half of 2023, sales of Ozempic and Wegovy rose by 58% and 363%, respectively. That’s after quarterly prescriptions for those types of GLP-1 treatments, which mimic a hormone in the gut to suppress a person’s appetite, increased 300% between early 2020 and the end of last year.
But as consumers and businesses pour more money and resources into tackling the obesity epidemic, which costs the U.S. more than $170 billion a year, drug developers aren’t alone in coming up with innovative solutions.
Signos, a five-year-old startup, is taking an approach that doesn’t involve pills.
The company is using off-the-shelf continuous glucose monitors, or CGMs, and providing real-time diet and exercise recommendations based on an individual’s readings. CGMs are small sensors worn on the upper arm that track glucose levels, primarily for people with diabetes. The information is wirelessly sent to a smartphone, allowing the user to better prevent emergencies.
Signos uses CGMs built by Dexcom. The startup has its own app that shows users how their body responds to specific foods, what causes their glucose to spike and when they should exercise to get the best results for weight loss.
On Tuesday, Signos said it closed a $20 million funding round led by Cheyenne Ventures and GV, formerly known as Google Ventures. Dexcom Ventures also contributed to the financing. Signos said it will use the fresh capital to continue its research into metabolic health and to expand its team, which is currently around 45 people.
“Whether you have five pounds to lose or 100, we want to make sure we’re able to help everybody,” Sharam Fouladgar-Mercer, Signos’ co-founder and CEO, told CNBC in an interview.
Customers who sign up for Signos can choose a one-month, three-month or six-month plan. With the half-year plan, users pay $143 a month, which includes all of the pricey CGMs they’ll need during that time. The company declined to share specific details about how many people are currently using its platform.
Fouladgar-Mercer said the long timelines are designed to attract users who are serious about their weight-loss journey. Additionally, the sensors themselves have a long wear time. The Dexcom G6 and G7, the latest devices, can measure glucose for up to 10 days. Signos currently supports the G6 and will soon work with the G7 as well.
Fouladgar-Mercer said Signos is using Dexcom’s CGMs as part of a clinical study approved by an institutional review board designated by the U.S. Food and Drug Administration to monitor biomedical research involving real people.
Fouladgar-Mercer said he created the company in 2018 partly because of his own struggle to manage weight throughout his life. He trained as an athlete and played hockey in college, but he said he noticed how food often affected him differently from the way it affected his teammates.
He said he always felt that, in an effort to understand an individual’s metabolism, there was a “critical component” missing, and it had been nagging at him for 30 years.
Signos helps users understand the right decision to make in the moment, but they can go “behind the scenes” and learn as much about the science as they’d like, Fouladgar-Mercer said. Users can also integrate sleep data, heart rate data, and exercise data from their Apple Watch to personalize their profile even more.
“Once they trust the system works and they understand the methodology, they can just follow the really quick, here’s what I do, here’s what I do, here’s what I do,” Fouladgar-Mercer said. “And that’s how you get behavioral change.”
Though Dexcom primarily develops its CGMs for patients with diabetes, the company is also working toward broader applications. For instance, next year it’s releasing a new product meant for people who aren’t taking insulin. Similarly, Abbott Laboratories, which dominates the global CGM market, is hoping to bring its first consumer-facing CGM, called Lingo, to the U.S. next year, adding personalized coaching with recommendations about diet, sleep and exercise.
Fouladgar-Mercer said Signos has more data points than “anybody does in the world for non-diabetics.” He added that since the company built its first product almost five years ago, it’s been able to focus on fine-tuning its technology.
“I don’t want to incorrectly set expectations,” Fouladgar-Mercer said. “I think a lot of times, it’s like, ‘Oh, lost X pounds in X days.’ That’s not what we’re trying to accomplish. It’s really, how do we put you on a sustainable journey? And that journey is not going to be done in two or three days.”
Fouladgar-Mercer said Signos can work well alongside Ozempic and Wegovy from Novo Nordisk and other GLP-1 treatments. Novo Nordisk’s share price has quadrupled since 2018, and the company is now the most valuable in Europe.
Fouladgar-Mercer said GLP-1 drugs are a “powerful tool” that can help people jump-start weight loss, but it can be challenging to keep weight off if they stop taking the medication. Platforms such as Signos can help to reinforce and maintain a healthier lifestyle over time, he said.
Ultimately, he said, he wants people to use Signos to learn how to make better choices that work best for their bodies.
Signos, Fouladgar-Mercer said, can use technology and data “to drive behavioral change, and then wrap that all in a system that really is focused on driving and solving this biggest problem we have in America, which is weight.”
WATCH: Novo Nordisk stops Ozempic trial early after signs of success
Weight loss drugs Wegovy, Ozempic may be linked to stomach paralysis
Still life of Wegovy an injectable prescription weight loss medicine that has helped people with obesity. It should be used with a weight loss plan and physical activity.
Michael Siluk | UCG | Getty Images
Blockbuster weight loss and diabetes drugs like Wegovy and Ozempic may be associated with an increased risk of three rare, but severe, stomach conditions in non-diabetic patients, according to a new epidemiological study released Thursday.
The study, published in the research journal JAMA, comes as Novo Nordisk‘s Wegovy, Ozempic and similar treatments skyrocket in popularity in the U.S. for their ability to cause dramatic weight loss over time. But those drugs, known as GLP-1s, are also facing increased scrutiny after some patients reported experiencing stomach paralysis and suicidal ideation while taking them.
GLP-1s work by slowing digestion to suppress a person’s appetite but can cause problems if that process slows down too much.
Researchers at the University of British Columbia said the conditions in the study include one disorder not named in the warning labels for those drugs: stomach paralysis, which slows or completely stops the movement of food from the stomach to the intestine and can cause symptoms like persistent vomiting.
The study also notes an increased risk of bowel obstruction, a disorder where food is blocked from passing through the small or large intestine, and pancreatitis, which refers to pancreas inflammation. The labels for the drugs already include warnings about pancreatitis and certain types of bowel obstruction.
The researchers specifically examined semaglutide – the active ingredient used in Wegovy and Ozempic – and another GLP-1 called liraglutide against another weight loss treatment called bupropion-naltrexone, which works differently to help patients lose weight. Wegovy is specifically approved for weight loss in the U.S., while Ozempic is only approved for diabetes.
Their research is the first large, population-level study to examine the risk of serious stomach conditions in non-diabetic patients specifically using GLP-1s for weight loss.
Previous studies have highlighted the risk of those conditions in diabetic patients taking GLP-1s, according to the researchers. People with diabetes are also at increased risk of experiencing stomach paralysis and pancreatitis overall, even without the treatments.
“That’s why we kind of wanted to take diabetes out of the equation,” said Mohit Sodhi, one of the authors of the study. “In addition to the fact that millions of people around the world are using these drugs to help them lose weight.”
A spokesperson for Novo Nordisk noted that some of the gastrointestinal side effects in the study are already included on the labels for its GLP-1s, adding that the company “stands behind the safety and efficacy of all of our GLP-1 medicines when used consistent with the product labeling and approved indications.”
“We recommend patients take these medications for their approved indications and under the supervision of a healthcare professional,” the spokesperson said. “Treatment decisions should be made together with a healthcare provider who can evaluate the appropriateness of using a GLP-1 based on assessment of a patient’s individual medical profile.”
Research findings
The study findings are based on an analysis of health insurance claim records for roughly 16 million U.S. patients.
Researchers specifically looked at people with a recent history of obesity who were prescribed semaglutide or liraglutide between 2006 and 2020. They excluded those with diabetes or patients who had been prescribed another diabetes drug.
Most patients in the study were prescribed liraglutide, but researchers said the increased risks they observed could apply to the entire GLP-1 drug class.
“Our data end date was the end of 2020, while the recent boom in semaglutide happened in the last year,” Sodhi said. “Nonetheless, we believe it is a class effect.”
CNBC Health & Science
Read CNBC’s latest health coverage:
The researchers measured the rate at which patients developed four different serious stomach conditions while taking semaglutide, liraglutide and bupropion-naltrexone, which are stomach paralysis, pancreatitis, bowel obstruction and biliary disease, a group of conditions affecting the gall bladder.
Compared with bupropion-naltrexone, GLP-1s were associated with a 9 times higher risk of pancreatitis, a 4 times higher risk of bowel obstruction and a more than 3 times higher risk of stomach paralysis, according to the study. The findings suggest the risks of those conditions are higher in patients specifically taking GLP-1s rather than other weight loss medications that work differently.
Around 7 out of every 1,000 patients experienced stomach paralysis while taking liraglutide, and nearly 10 out of every 1,000 patients experienced that condition while taking semaglutide.
And Sodhi noted, “the number just continues to climb when you blow it up to the population level.”
“When you have more than a million people taking the medication worldwide, that’s 10,000 people who could potentially experience gastroparesis according to the incidence rate for semaglutide,” he told CNBC. “It’s rare, but that’s still a lot of people.”
This recent archive picture, taken 17 August 2023 and distributed Tuesday 26 September 2023, shows a package with Ozempic medicine at a hospital in Bonheiden. T
Dirk Waem | AFP | Getty Images
Almost 5 out of every 1,000 patients experienced pancreatitis while taking semaglutide, while roughly 8 out of every 1,000 patients experienced that condition while taking liraglutide.
Meanwhile, around 8 out of every 1,000 patients experienced bowel obstruction while taking either of those GLP-1s.
The researchers also found a high rate of biliary disease in patients taking either liraglutide or semaglutide, but they said the difference was “not found to be statistically significant.”
The researchers hope the study will inform health-care providers prescribing GLP-1s about the potential drawbacks of taking the drugs.
“We’re all big proponents for informed patient consent,” Sodhi said. “If someone has decided they would like to take a GLP-1 for weight loss, we encourage them to have a conversation with their provider about how it may help them achieve their goals. But they should also be made aware of the potential drawbacks of taking this medication.”
So much for sleeping in: Study says morning exercise is best for losing weight.
If you want to lose weight, you may be better off hitting the gym earlier in the day.
That’s the finding from a new study published in the research journal Obesity. The study examined data from more than 5,000 participants in a 2003-06 U.S. health survey and looked at their workout patterns. The results showed that the “morning cluster” exercisers had a lower body mass index and waist circumference than their counterparts who exercised in the middle of the day and in the evening.
Authors…
I’ve been testing Apple’s two new Pro iPhones for the past several days. They hit store shelves on Friday.
The two Pro iPhones are significant year-over-year upgrades, with improvements to the camera, an improved physical body, and a new button. Most important for me, the high-end models are much lighter in weight and really make a big difference during daily use. I’d recommend them to any iPhone user who’s ready to upgrade, or anyone who has complained about how big and heavy smartphones have gotten.
They’re not cheap.
The 6.1-inch iPhone Pro model starts at $999 for 128GB of storage and goes up to $1499 for 1TB. I’d recommend the $1099 model with 256GB of storage for most people. The bigger phone, the 6.7-inch iPhone Pro Max, now starts at $1199, $100 more than last year, but the entry-level model has 256GB of storage and maxes out at 1TB for $1599.
In addition to the Pro models, Apple also offers two cheaper options, the iPhone 15 and iPhone 15 Plus, which cost $799 and $899 and up. They have the chip from last year’s iPhone 14 Pro, some camera improvements, and Apple’s “dynamic island” that hides the front-facing cameras. Most people will be perfectly happy with the mainstream iPhone 15 models, which are close to state-of-the-art.
But Apple’s Pro phones have the newest chip, the A17 Pro, a zoom lens, and a display with a faster refresh rate. For people who are picky about their personal technology, the pro models are significantly more desirable than the mainstream models, and their improvements often trickle down Apple’s lineup in the coming years.
Here’s what’s new and exciting about the iPhone 15 Pro phones:
What’s good
The “natural titanium” finish is the most noticeable new iPhone color. This is also a close look at the new Action Button, which replaces the mute switch.
Kif Leswing/CNBC
This year’s Pro iPhones have one visible and welcome change: They use titanium for the exterior frame, instead of stainless steel.
One color, “natural titanium,” shows off the new metal, but withmost other colors, it isn’t as immediately noticeable. But while the iPhone 15 Pro might look the same as last year’s model from a distance, it’s actually the biggest change to the iPhone’s exterior in years.
Titanium is a huge upgrade for daily use because it makes the phone lighter. It feels better in the hand. Apple’s Pro phones have been getting heavier since 2019, but this phone reverses the trend.
Apple says that the 6-inch iPhone 15 Pro is 187 grams, or 9% lighter than last year’s model. The iPhone 15 Pro Max, with a bigger 6.7-inch screen, weighs 8% less. But in practice, it feels like an even bigger reduction. Going back to my old iPhone 14 Pro, it feels like a brick. The weight difference between this year’s Pros and last year’s is noticeable even with a case.
Not only are this year’s Pro phones lighter, but they are also slightly narrower and shorter in length, although they’re marginally thicker than last year’s devices. They have a smaller border (bezel) around the same-sized screen, and it’s noticeable when holding the two devices side-by-side. But this also means last year’s cases won’t work with this year’s phones.
The iPhone 14 Pro is in the back and the iPhone 15 Pro is in the front. This year’s model has slighly smaller bezels. It’s subtle but noticable.
Kif Leswing/CNBC
The weight difference is subtle but will be meaningful to many people, making daily use more delightful — and marathon TikTok and YouTube sessions less tiring.
I used the iPhone 15 Pro without a case, and it didn’t feel particularly fragile. But phones drop and break, and the new frame comes with one additional benefit: Significantly cheaper repairs. If the back glass on the iPhone 15 shatters, Apple is charging either $169 or $199 for a replacement, depending on screen size. On the iPhone 14 Pros, that price was either $499 or $549. Apple says it’s because this model is much easier to repair.
Zoom lens
Apple has also improved the zoom camera on the iPhone 15 Pro Max.
While the smaller phone still has a 3x zoom, the bigger phone now comes with a 5x zoom lens that mimics some of the better physical zoom lenses on standalone cameras that can be as long as 10 inches. Basically, the new iPhone allows you to get closer to your subject without physically getting closer.
On the left, an iPhone 15 Pro Max photo taken at 5x zoom. On the right, an iPhone 15 Pro photo taken with 3x zoom.
CNBC/Kif Leswing
It also comes with a nifty new kind of three-dimensional image stabilization that prevents the zoom shots from getting blurry, which is a problem with actual big zoom lenses. The handheld zoom shots on the iPhone 15 Pro Max were pretty sharp and clear.
The new lens will be very useful for day-to-day usage, as it makes more of the world around you easier to photograph. Parents will use it for kids’ sports, outdoors enthusiasts will use it take photos of birds and wildlife, and tourists will use it to get better vacation shots.
On the left: 2x zoom photo taken with the iPhone 15 Pro Max. On the right: 5x zoom photo taken with the iPhone 15 Pro Max
Kif Leswing/CNBC
It’s also a reason to reach for the bigger iPhone 15 Pro Max over the cheaper but lighter and smaller iPhone 15 Pro.
On the left: 2x zoom taken with iPhone 15 Pro Max. On the right: 5x zoom taken with iPhone 15 Pro Max.
The same photo of San Francisco’s ferry building, without zoom, through the iPhone 15 Pro Max main camera.
Kif Leswing/CNBC
USB-C
Apple’s iPhones have a new charger for the first time in a decade. Meet the USB-C port.
Behold: The USB-C port that will be on all iPhones going forward.
Kif Leswing/CNBC
Apple’s change was prompted by European regulation, and will make being an iPhone owner a little more convenient, because you’ll be able to share your iPhone cord with laptops, headphones, iPads, and other newer gadgets. You can use a laptop charger to charge the iPhone, or use your iPhone’s charger to fill up your headphones.
The port change on the iPhone Pros also opens up some cool new abilities. For example, if you have a USB-C monitor dock, like many corporate workers do, you can now likely plug your phone into a monitor, which mirrors what’s on your phone screen, for presentations, or perhaps to watch a movie or play a game. That was possible before, with a dongle, but now anyone with a new iPhone and recent monitor dock can do it.
You can also now use a powered-up iPhone to charge other gadgets, such as AirPods or Apple Watch, or even another iPhone, using a cord coming from its USB-C port. They don’t charge fast, but it works.
The new USB-C port now even allows iPhones to charge each other, or AirPods, or other gadgets. When two USB-C iPhones are plugged together, the one with less charge automatically charges. Works with last year’s Lightning port too, but they always charge, and never charge other gadgets.
Kif Leswing/CNBC
USB-C is a great improvement, although it won’t be immediately smooth for a lot of people. For example, the cord in my car for Apple CarPlay is currently using Lightning, the old connector, and not everyone in my family is upgrading to the new phones. I’ll have to use a dongle for a while.
Action button
The other big physical change to the iPhones this year is the Action button, which replaces the mute-switch.
The button is surprisingly nice, and I can see a lot of people using it every single day. Most people leave their phones on mute all the time, and there are several ways to mute or unmute your phone through software settings.
Apple’s action button can be set to different use cases in the Settings app, with a surprisingly sophisticated 3D interface.
Screenshot/CNBC
The new button provides haptic feedback when it’s pressed, and Apple has provided several convenient ways to use it.
My personal favorite is as a camera button. Press it quickly, and it brings up the camera. Press it again, and it takes a photo. If you hold it down, it takes a video.
Another interesting application is to quickly bring up a translation app that can take your spoken words and turn it into one of several foreign languages, and it translates responses as well.
The button is also customizable through Apple’s iPhone macro program called Shortcuts, which means people will come up with a number of creative uses once it’s released to the public.
Nice to have
New portrait features
Other camera improvements include the ability to change focus in many photos after they’re taken. It’s part of the ability to use “Portrait Mode” without specifically taking a Portrait Mode photo. Any photo taken with the iPhone 15 Pro’s camera that either has a manual focus (tapping the screen) or has a human or pet in the frame will automatically capture the depth information needed to tweak the photo after-the-fact.
Photos with Apple’s depth data can do lots of stuff, like changing the focus in the image, or Portrait Mode, which can vignette the background or give a softer focus to the subject. Photos with Apple’s depth mdata are marked in the camera roll with a stylized “f” symbol.
These screenshots show a photo taken outside of portrait mode. In the left two panels, you can see how you can shift the focus in the photo after it was taken. The right photo is how it looked right after it was taken.
Screenshot/CNBC
Apple’s main camera on the Pros also takes 24 megapixel photos, combining a larger 48-megapixel image with a smaller 12-megapixel image to synthesize a new photo with better range of light and additional details. It’s a subtle improvement but comforting if you shoot a lot of photos and just want them all to be good.
A17 chip
The iPhone Pros get the A17 Pro chip, the latest and greatest Apple silicon, as compared to the mainstream iPhone 15s, which have last year’s Pro model A16 Bionic chip.
In day-to-day use, you won’t notice the difference, because the iPhone is generally snappy. But Apple says the chip has a new 6-core GPU design that can handle “ray tracing,” an advanced kind of graphics computation previously limited to high-end gaming cards that essentially simulates light. It’s also got a better “neural engine” for running artificial intelligence.
There aren’t ray-tracing enabled games available yet on Apple’s App Store, but there are console-level titles such as the forthcoming Resident Evil Village that will take advantage of the new chip.
For most people, having the latest chip means there’s another year at the end of the phone’s life when it’s likely to get updates.
Standby mode
Apple’s Standby mode turns your iPhone into an updating dashboard that users can customize.
Kif Leswing/CNBC
One of the phone’s niftiest new features allows it to turn into a dashboard that displays real-time information, such as calendar appointments, photos, and even things like how close your pizza is to being delivered. This feature works with older phones, too, that have updated to the latest version of iOS, and requires a MagSafe charging stand. It’s going to be great for office workers.
Still some quirks
Heat
I only encountered a hot iPhone 15 Pro Max once, when playing the Apple Arcade title “Zookeeper World.” However, for about 5 minutes, the phone was too hot to hold, especially on the titanium frame, and I had to put it down. Heat issues haven’t cropped up since in my testing.
iPhone setup
Apple’s automatic iPhone transfer system has come a long way. When I got my review unit, all I had to do is sign in with my Apple ID, and hold my old phone next to the new one, and my texts, apps, photos and files all transferred. I’m an iCloud subscriber, but it was still pretty seamless.
But the process still isn’t perfect, and users should know it may take a day to make sure everything is transferred over. For example, my Apple Watch didn’t immediately transfer to pairing with the new phone — and I had to be careful, because I had a farecard on my Watch with a significant amount of money on it, and if I wasn’t careful about how I transferred it, I might have lost it. A few health settings didn’t immediately sync either, and required my attention to sort out.
Conclusion
The iPhone 15 Pro on the left, the iPhone 15 Pro Max on the right.
Kif Leswing/CNBC
The iPhone 15 Pros are one of the best upgrades in many years for Apple’s high-end phones, although the core of what an iPhone does hasn’t changed at all. Any of Apple’s phones currently on sale — which include 2022 and 2021 models at discounts — can make calls, send texts, download apps, and take great photographs. If you’re upgrading after a few years, all of Apple’s phones are big improvements, and they’ll all get the job done.
But there are also people who know they use their phone for hours per day. In fact, Apple’s Screen Time feature often reminds them of just how much time each week they spend on their iPhone.
For these people, even if they have last year’s phone, it’s worth going to an Apple Store or Best Buy, picking up the new Pro devices, and seeing if they like the decreased weight and new balance. I think the lighter, more wieldable phone is a major improvement in the phone’s physical form, even if it looks largely the same as last year’s models.
The other question is whether to pay the extra $200 to get the Pro Max when it’s got the same chip and main features as the $999 iPhone 15 Pro. The Pro Max is still a big phone with a 6.7-inch screen, but it’s much lighter this year, and the 5x zoom is a significant feature and something worth getting for people who take a lot of photos, like parents or photographers.