U.S. small-cap stocks are “challenged” as Wall Street pushes out expectations for when the Federal Reserve may begin lowering interest rates due to concerns over persistent inflation, according to BofA Global Research.
Source link
expectations
After a six-week run, altcoins are cooling off, leading some to fear the worst. However, in a post on X, one crypto analyst urges calm, saying the dip is a normal part of any bull market and may even present opportunities for discerning traders and investors.

Altcoins Drop, Follow Bitcoin
After posting solid gains and rallying to as high as $73,800 on March 13, Bitcoin edged lower on March 14 and continues under pressure when writing. Considering the positive correlation between Bitcoin and altcoins, leading altcoins like Ethereum, XRP, Cardano, and Solana retraced slightly.
Related Reading: Bitcoin Crash Triggered By Failed $1 Billion Hedge Fund Spread Trade: Expert
When writing, Ethereum is far from the $4,090 recorded last week. At the same time, the Solana uptrend has slowed, with $200 proving elusive for optimistic traders. Meanwhile, XRP, despite the high optimism when prices roared higher on March 11, is struggling for momentum and is currently trending lower.
While the altcoin has been drawn down, the total crypto market cap drop and fear creep in have caused the analyst to remain bullish. Despite the correction, the altcoin uptrend remains, and most coins, including Ethereum, Solana, and Cardano, could post more gains.
Ethereum, Solana, and BNB Fundamentals Are Solid
To illustrate, the crypto community remains upbeat because of Dencun and its potential to enhance Ethereum and the broader layer-2 ecosystem. Ethereum rollup platforms, including Base and Optimism, that have activated Dencun features have seen decentralized applications (dapps) launching on their platforms benefit.
Uniswap Labs, the team behind Uniswap, a leading decentralized exchange, reported that transaction fees on users trading on Optimism have fallen even further after activating Dencun. This reduction in fees may be a catalyst to drive more activity on-chain, propping up prices.
On the other hand, Solana bulls are upbeat. Since September 2023, SOL has been on a sharp uptrend. At spot rates, the coin roared to register fresh 2024 highs.
With this performance, Solana outperforms Bitcoin and other altcoins, including ETH. CoinMarketCap data shows that SOL has risen by 15% in the past week, closely trailing BNB, the native currency of the BNB Chain ecosystem.
Analysts are looking at the upcoming Firedancer upgrade to fortify Solana’s infrastructure further and increase reliability. Meanwhile, regulatory developments in the United States are evolving positively, propping up altcoins, mainly BNB.
Overall, the crypto uptrend remains even with prices cooling off. More importantly, even with falling prices, sentiment remains bullish. In the past week, CoinMarketCap data shows that the Crypto Fear and Greed Index is at “extreme greed” territory.
Feature image from Shutterstock, chart from TradingView
Disclaimer: The article is provided for educational purposes only. It does not represent the opinions of NewsBTC on whether to buy, sell or hold any investments and naturally investing carries risks. You are advised to conduct your own research before making any investment decisions. Use information provided on this website entirely at your own risk.
Bristol Myers Squibb results top expectations as newer drugs help fuel growth

Bristol Myers Squibb Co.
BMY,
on Friday reported fourth-quarter results that beat analysts’ expectations, helped by portfolio stalwarts like the blood thinner Eliquis as well as strong growth among newer drugs.
The drugmaker reported fourth-quarter net income of $1.762 billion, or 87 cents per share, down from $2.022 billion, or 95 cents per share, in the year-earlier period. Adjusted earnings per share came to $1.70, down 7% from a year earlier but ahead of the FactSet consensus of $1.55. Revenue totaled $11.477 billion in the quarter, up 1% from a year earlier and topping the FactSet consensus of $11.192 billion.
For the full year 2024, Bristol Myers Squibb said it expects sales growth in the low single-digits and adjusted earnings per share of $7.10 to $7.40, ahead of analysts’ estimates.
Among Bristol Myers’ top-selling products, growth in sales of blood thinner Eliquis and cancer drug Opdivo helped offset declining sales of multiple myeloma treatment Revlimid, which is facing generic competition.
Eliquis, one of 10 drugs selected for the first round of Medicare price negotiations under the Inflation Reduction Act, generated $2.87 billion in sales in the fourth quarter, up 7% from a year earlier and slightly ahead of analysts’ expectations.
Revlimid sales fell 36% from a year earlier, to $1.45 billion in the fourth quarter.
Bristol Myers’ new product portfolio accounted for $1.072 billion in sales in the quarter, up 66% from a year earlier. Melanoma treatment Opdualog and anemia drug Reblozyl helped fuel that growth, but sales of the CAR-T cell therapy Abecma fell 20%, to $100 million.
The company’s partner on Abecma, 2seventy bio Inc.
TSVT,
said earlier this week that it is pivoting to focus exclusively on the development and commercialization of the cell therapy and is selling its research-and-development pipeline to Regeneron Pharmaceuticals Inc.
REGN,
With analysts watching for a reset after a rough 12 months, as the shares have dropped more than 30%, Bristol Myers has been on a shopping spree lately. In December, the company struck deals to buy radiopharmaceuticals company RayzeBio Inc.
RYZB,
as well as Karuna Therapeutics Inc.
KRTX,
which has a key experimental schizophrenia treatment, KarXT. A regulatory decision on KarXT is expected in September.
Last month, the company closed its acquisition of cancer drugmaker Mirati Therapeutics Inc.
Bristol Myers Squibb shares are down 5.2% year to date, while the S&P 500
SPX
has gained 2.9%.
George Frey | Bloomberg | Getty Images
Weight loss drugs exploded into the public eye this year, and 2024 will bring more change to the evolving market.
The drugs skyrocketed in popularity in 2023 as they helped patients shed significant weight, despite hefty price tags, mixed insurance coverage and a handful of unpleasant side effects.
Demand for the drugs is unlikely to slow down in 2024, especially as treatments gradually become more accessible. Much of Wall Street believes the weight loss drug market will only expand, with some analysts projecting that it will be worth $100 billion by the end of the decade. Goldman Sachs analysts expect 15 million U.S. adults to be on obesity medications by 2030.
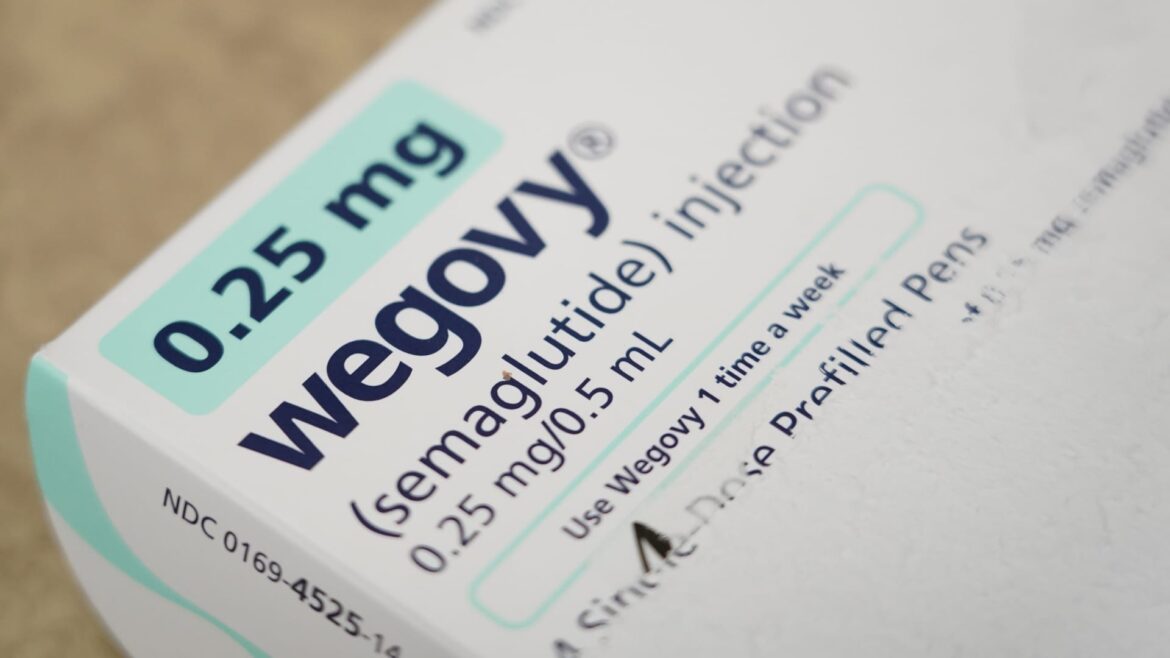
But next year, investors will be watching how the dominant players in the market, Novo Nordisk and Eli Lilly, navigate supply issues plaguing their treatments. Patients have been struggling to get their hands on Novo Nordisk’s weight loss injection Wegovy, its diabetes treatment Ozempic, and Eli Lilly’s diabetes injection Mounjaro.
Analysts expect supply constraints to improve but note that the broader issue will take years to resolve.
Outside of supply headwinds and the lack of broader insurance coverage for weight loss drugs, Novo Nordisk and Eli Lilly have a big year ahead of them.
Novo Nordisk could win approvals for expanded use of Wegovy in the U.S. and Europe. Eli Lilly’s newly approved weight loss drug, Zepbound, could garner more than a billion dollars in sales in its first year on the market.
Both companies are also expected to release new data that could show other potential health benefits of their drugs beyond weight loss and diabetes management, which may increase insurance coverage down the line.
Next year may mean even more to the other companies hoping to join what’s so far been a two-horse race to make weight loss treatments.
New drug data from Pfizer and Amgen, and the potential for more buyouts or collaborations between larger companies and smaller makers of obesity drugs, could alter the market’s competitive landscape in the coming months.
Supply issues could ease but won’t go away
Some analysts said supply constraints will likely persist for years, but expect them to ease in 2024 as Novo Nordisk and Eli Lilly work to expand manufacturing capacity for their drugs.
Novo Nordisk during its third-quarter earnings call in November said it is “looking at significantly scaling our supply” of Wegovy in the U.S. in 2024. TD Cowen analyst Michael Nedelcovych told CNBC that the company during the call appeared to suggest that such a change wouldn’t look like a big jump in supply but rather steady improvements over time.
Supply could increase more significantly years from now: Novo Nordisk in November said it would invest $6 billion to expand its manufacturing facilities in Denmark, noting it will finish construction from the end of 2025 through 2029. The company also said it would spend around $2.3 billion to expand another production site in France.
Top weight loss and diabetes drugs
Wegovy from Novo Nordisk is a weekly weight loss injection for adults with obesity or who are overweight. The drug mimics a hormone produced in the gut called GLP-1 to suppress a person’s appetite.
Zepbound from Eli Lilly is a weekly weight loss injection for adults with obesity or who are overweight. The treatment mimics GLP-1 and another gut hormone called GIP to reduce appetite and food intake.
Ozempic from Novo Nordisk is a weekly injection that helps lower blood sugar levels in adults with Type 2 diabetes. The medication mimics GLP-1 to suppress appetite and help the pancreas make more insulin.
Mounjaro from Eli Lilly is a weekly injection that helps lower blood sugar levels in adults with Type 2 diabetes. The drug mimics GLP-1 and GIP to curb appetite and stimulate insulin production.
Meanwhile, Eli Lilly said during its third-quarter earnings call in November that supply of Mounjaro has improved in the U.S. even as it remains constrained around the globe.
Executives also said that Eli Lilly is on track to achieve its goal of doubling production capacity for drugs such as Mounjaro, in part through investments in new manufacturing sites in North Carolina and Indiana.
But Eli Lilly CEO David Ricks said on the call that the company is “aggressively planning” further production buildup for Mounjaro and other drugs. He added that “it’s a problem we work on every day. So we’re not at all happy with the capacity.”
Zepbound could become a blockbuster
The FDA approves Eli Lilly’s Zepbound, a weight loss drug similar to Ozempic and Wegovy.
Courtesy: Eli Lilly
Morgan Stanley expects Zepbound to rake in $2.2 billion in sales in 2024, according to a note released after the drug’s approval in November. Meanwhile, Bank of America analysts in a November note projected $2.7 billion in Zepbound revenue in 2024.
Some analysts expect far more sales growth for Zepbound and Mounjaro beyond 2024. Tirzepatide, the active ingredient in both drugs, has a “very strong shot of being the best-selling molecule of all time in the pharmaceutical industry,” said Guggenheim’s Fernandez.
Wall Street is enthusiastic about Zepbound in part because it may cause more weight loss than Wegovy. Studies directly comparing the two, including an ongoing trial from Eli Lilly, would need to confirm that.
Results from that trial could come out next year after initial data from separate studies examining Zepbound as a potential treatment for other health conditions, including heart failure.
Mixed insurance coverage will likely weigh on sales of Zepbound and other weight loss drugs in 2024, but Eli Lilly has already secured some coverage for the drug.
Wegovy could make history again
Wegovy made history this year when it slashed the risk of serious heart problems by 20% in people with obesity and heart disease in a late-stage trial. In 2024, the drug could shake up the pharmaceutical industry again if U.S. and European regulators decide to approve it for that purpose.
Those potential approvals, which would make Wegovy the first GLP-1 drug to have an expanded use for heart health, are a “foregone conclusion” for Novo Nordisk, Cantor Fitzgerald’s Louise Chen told CNBC.
Still life of Wegovy an injectable prescription weight loss medicine that has helped people with obesity. It should be used with a weight loss plan and physical activity.
Michael Siluk | UCG | Getty Images
An FDA approval could potentially increase the uptake of Wegovy, encouraging more obesity specialists, primary care providers and cardiologists to prescribe it to eligible patients, said Dr. Eduardo Grunvald, medical director for UC San Diego’s Center for Advanced Weight Management.
An approval may also put more pressure on U.S. insurers to eventually cover Wegovy and similar weight loss treatments, opening the door for broader use.
Eli Lilly is also studying the cardiovascular benefits of Zepbound in a phase three clinical trial in diabetes patients with increased cardiovascular risk, and results are expected in late 2024. The drugmaker is conducting a similar study in obese patients with heart-health risks, but results may not come until 2027.
Meanwhile, Novo Nordisk’s other treatments could reach their own milestones next year.
Novo Nordisk expects to release data in the first half of 2024 from a late-stage trial examining Ozempic as a treatment for kidney failure in diabetes patients with chronic kidney disease. The company hinted that the trial would be a success when it halted the study a year earlier than planned in October based on an interim analysis.
Upcoming clinical trial data releases
- A phase-three trial from Eli Lilly on Zepbound as a treatment for cardiovascular complications in diabetes.
- A phase-three trial from Novo Nordisk on Ozempic as a treatment for kidney failure in diabetes patients with chronic kidney disease.
- A phase-three trial from Novo Nordisk on a 25-milligram version of its once-a-day weight loss pill.
- A phase-three trial on Zepbound as a potential treatment for heart failure in patients with obesity.
- A phase-three trial on Zepbound as a potential treatment for non-alcoholic fatty liver disease, which is caused by fat buildup in the liver, in patients with obesity.
- A phase-three trial on Zepbound as a potential treatment for obstructive sleep apnea, or the pause of breathing during sleep due to blocked airways, in patients with obesity.
- A phase-three trial on IcoSema, a combination of once-weekly insulin and once-weekly semaglutide, in patients with diabetes.
Novo Nordisk will also release phase three clinical trial data on a 25-milligram version of its once-a-day weight loss pill, which uses semaglutide, the same active ingredient as in Ozempic and Wegovy.
That trial is crucial because Novo Nordisk is waiting to see that data before filing for approval of the oral weight loss drug, said Cowen’s Nedelcovych. He added that in the long term, the availability of weight-loss pills could boost capacity for their injectable counterparts.
Also in 2024, a study following patients from a previous late-stage trial could potentially generate data supporting Wegovy as a treatment for preventing the development of diabetes, Nedelcovych said.
A make-or-break year for Pfizer
New data next year will be crucial to determining whether Pfizer gets a piece of the weight loss drug space. The stakes are high: CEO Albert Bourla has said the company hopes to capture $10 billion of that market.
Pfizer axed a twice-daily version of the only obesity product in its pipeline earlier this month after patients taking the pill lost significant weight but had trouble tolerating the drug in a mid-stage study.
Now, the company is pinning its hopes on a once-a-day version of the pill, known as danuglipron, which it believes may cause fewer adverse side effects. Pfizer said it expects to release more data on that version of the drug in the first half of 2024, which will help the company decide whether to start a late-stage study on the pill.
CFOTO | Future Publishing | Getty Images
However, some analysts have raised questions about whether the once-a-day version will be easier to tolerate.
“Despite ongoing work, tolerability still appears to be an issue with the product, and it is not clear to us why this will improve” in a phase three trial or with a once-daily version, JPMorgan analyst Chris Schott said in a December note.
Barclays analyst Carter Gould said in a December note that it is “increasingly apparent” that the company will have to look externally for an obesity treatment, whether that’s through an acquisition or partnership, to capture a slice of the weight loss drug market like it had hoped.
Meanwhile, upcoming data will reveal how serious Amgen’s weight loss drug portfolio is. In the first half of 2024, Amgen is slated to publish early stage trial data on an oral weight loss medication.
In the second half of the year, Amgen plans to release mid-stage trial data on an injectable drug that helped cause up to 14.5% weight loss after 12 weeks in an early study.
Watch for buyouts and partnerships
Pfizer isn’t the only company that could benefit from looking externally for obesity drugs.
Larger drugmakers used acquisitions of smaller businesses, or partnerships with them, to carve out space in the weight loss drug market this year. More companies could deploy the strategy next year, analysts said.
“There are a bunch of other large-cap pharmas on the list who could do this,” said Cantor Fitzgerald’s Chen.
Swiss company Roche said earlier this month it would buy the privately held U.S. obesity drugmaker Carmot Therapeutics for $2.7 billion. AstraZeneca signed a licensing agreement with Chinese biotech company Eccogene to develop an obesity pill.
Novo Nordisk and Eli Lilly have also snapped up smaller obesity drug companies this year to maintain their dominance in the market.
Recent weight loss drug buyouts and partnerships
- Roche in December said it will buy privately held obesity drugmaker Carmot Therapeutics for $2.7 billion.
- AstraZeneca in November said it signed a licensing agreement with Chinese biotech company Eccogene to develop an obesity pill.
- Novo Nordisk in August said it will acquire the privately held obesity drugmaker Inversago Pharma for $1.08 billion.
- Novo Nordisk in August said it will acquire Embark Biotech, which develops obesity and diabetes drugs, for up to $500 million.
- Eli Lilly in July said it will acquire privately held obesity drugmaker Versanis for $1.93 billion.
In a statement to CNBC, Novo Nordisk said it has increased its focus on “sourcing and elevating external innovation” to complement its in-house products and broaden its drug pipeline, especially for diabetes, obesity, cardiovascular disease and rare blood disorders.
The company also said it is interested in the “full range of business development activities,” from acquisitions to partnerships on early or late-stage products, when it comes to companies with new biological drugs, new potential treatment targets and new mechanisms of action, or how a drug works.
Chen said acquisitions or partnerships may be the only way for small- to mid-cap weight loss drugmakers to catch up with Eli Lilly and Novo Nordisk.
Some smaller companies have indicated that they are open to the idea: Altimmune said Dec. 5 that it is looking for partners to launch and develop its experimental obesity drug pemvidutide.
Shares of Altimmune have jumped more than 140% since Nov. 30, when the company released mid-stage trial data showing that its injectable drug caused 15.6% weight loss on average after 48 weeks.
Other smaller weight loss drugmakers include Structure Therapeutics, whose once-daily pill helped overweight or obese patients lose up to 10 pounds of weight on average after a month in an early-stage trial. The company is expected to report mid-stage trial data on its drug in diabetes patients this month and more results on the pill in patients with obesity early next year, Guggenheim’s Fernandez noted.
Still, some bigger drugmakers may wait to see larger and later-stage data from smaller companies before moving to acquire them. That data may not come out until 2025 or later for many firms, said Fernandez.
Chainlink Staking Program Exceeds Expectations, Drives LINK Price Up By 12%
In a significant development for the blockchain data-oracle project, Chainlink (LINK) has witnessed a significant response to its enhanced crypto-staking program, amassing over $632 million worth of its LINK tokens within a remarkably short period.
The company announced a recent press release highlighting the “overwhelming demand” during the early-access period, which filled the staking limit in just six hours.
Chainlink Unveils Staking v0.2
Chainlink, recognized as the industry-standard decentralized computing platform, unveiled Chainlink Staking v0.2, the latest upgrade to the protocol’s native staking mechanism.
The Early Access phase has commenced, inviting eligible participants to stake up to 15,000 LINK tokens. This phase will last four days before transitioning into the General Access phase, enabling investors to stake up to 15,000 LINK tokens as long as the staking pool remains unfilled.
Per the announcement, the upgrade introduces an expanded pool size of 45,000,000 LINK tokens, equivalent to 8% of the current circulating supply. This enlargement aims to enhance the accessibility of Chainlink Staking, enabling a more diverse audience of LINK token holders to participate.
Staking forms an integral part of Chainlink Economics 2.0, which brings an additional layer of cryptoeconomic security to the Chainlink Network. Specifically, Chainlink Staking empowers ecosystem participants, including node operators and community members, to support the performance of Oracle services by staking LINK tokens and earning rewards for contributing to network security.
While v0.1 served as the initial phase of the Staking program, v0.2 has been restructured into a fully modular, extensible, and upgradable Staking platform. Building upon the lessons learned from the previous release, the v0.2 beta version focuses on several key objectives.
Chainlink is introducing several new features to enhance its staking program. These include a new unbinding mechanism that provides more flexibility for Community and Node Operator Stakers.
Additionally, security guarantees for Oracle services are being reinforced by slashing node operator stakes. A modular architecture is being adopted to support future improvements and additions, and a dynamic rewards mechanism is being introduced to seamlessly accommodate new external sources of rewards in the future, such as user fees.
Following the conclusion of the Early Access phase on December 11, 2023, the v0.2 staking pool will transition to General Access. At this stage, anyone will have the opportunity to stake up to 15,000 LINK tokens.
LINK Surges To New Yearly High
Given Chainlink’s successful upgrade, LINK, the native token of the decentralized computing platform, experienced a significant surge of 12%, reaching a price as high as $17.305.
This price level has not been seen since April 2022, signifying a new yearly high for the cryptocurrency. However, LINK has retraced slightly and is currently trading at $16.774.
Crypto analyst Ali Martinez has highlighted a critical support zone for Chainlink. Martinez noted that over 17,000 addresses purchased 47 million LINK tokens from $14.4 to $14.8.
This accumulation by many addresses suggests strong buying interest in this price range, potentially acting as a support level for the token.
While the support zone may hold and trigger a rebound in the price of LINK, Martinez cautions that investors should remain vigilant. Any signs of weakness, such as a breach of the support zone or negative market sentiment, could prompt investors to sell their LINK holdings to avoid losses.
It remains to be seen whether LINK can maintain its position above these critical levels and whether the broader cryptocurrency market will enter an accumulation phase or experience a retracement after the significant upward movement witnessed in recent weeks.
Such a retracement could potentially impact LINK’s price and lead to a test of the support above levels. On the other hand, the token faces immediate resistance at $17.483, $18.069, and $18.910. These represent the final hurdles to overcome before LINK reaches the $20 milestone.
Featured image from Shutterstock, chart from TradingView.com
UiPath Stock Soars 27% as AI-Powered Earnings and Revenue Beat Expectations
Shares of UiPath (PATH 26.72%) rocketed 26.7% on Friday, following the enterprise automation software company’s release on the prior afternoon of a better-than-expected report for the third quarter of fiscal 2024 (ended Oct. 31).
The stock’s steep rise is attributable to revenue and adjusted earnings exceeding Wall Street’s estimates, with the bottom-line beat a sizable one. Fourth-quarter revenue guidance, which was slightly stronger than analysts had expected, likely also provided a tailwind for the stock.
UiPath’s key numbers
| Metric | Fiscal Q3 2023 | Fiscal Q3 2024 | Change |
|---|---|---|---|
| Revenue | $262.7 million | $325.9 million | 24% |
| GAAP operating income | ($67.0 million) | ($55.8 million) | Loss narrowed 17% |
| GAAP net income | ($57.7 million) | ($31.5 million) | Loss narrowed 45% |
| Adjusted operating income | $18.0 million | $43.7 million | 143% |
| Adjusted net income | $26.7 million | $69.1 million | 159% |
| GAAP earnings per share | ($0.10) | ($0.06) | Loss narrowed 40% |
| Adjusted EPS | $0.05 | $0.12 | 140% |
Data source: UiPath. GAAP = generally accepted accounting principles. Fiscal Q3 2024 ended on Oct. 31, 2023.
Investors should focus on the adjusted numbers because they exclude one-time items.
Wall Street was looking for adjusted EPS of $0.07 on revenue of $315.6 million, so UiPath easily exceeded both expectations.
GAAP gross margin was 85%, up from 84% in the year-ago period. Adjusted gross margin was 87%, up from 86% in the third quarter of fiscal 2023.
In fiscal Q3, UiPath generated cash of $42 million running its operations, bringing its fiscal-year-to-date total cash flow from operations up to $153.5 million. This is a huge improvement from the first nine months of the prior fiscal year when the company used $104 million running its operations.
The company ended the period with cash, cash equivalents, and marketable securities of $1.82 billion, up from $1.76 billion in the year-ago quarter.
What happened with UiPath in the quarter?
- License revenue was $148.1 million, up 25% year over year.
- Subscription services revenue landed at $167.5 million, up 29%.
- Professional services and other revenue was $10.3 million, down 28%.
- Annualized renewal run-rate (ARR) grew 24% year over year to nearly $1.38 billion.
- Dollar-based net retention rate was 121%. This means that existing customers expanded their spending on the company’s services by an average of 21% over the last year. This is a very good result and suggests that customers are finding value in UiPath’s services.
What management had to say
Here’s what Co-CEO Ron Enslin had to say in the earnings release:
I am pleased with our strong third quarter results with ARR [annualized renewal run-rate] growing 24 percent year-over-year to $1.378 billion, driven by the team’s execution and the transformational results we deliver. My conversations with customers and partners validate the strategic role enterprise automation plays in digital transformation and I am excited about the investments we continue to make in AI [artificial intelligence] to further extend our market leadership.
Guidance
For the fourth quarter of fiscal 2024, management guided for revenue in the range of $381 million to $386 million. This would represent growth of 24% to 25% year over year. At the midpoint ($383.5 million), the outlook slightly exceeds the $382 million Wall Street had been projecting.
Moreover, the company expects the quarter’s adjusted operating income to be about $78 million, which would represent growth of approximately 13% year over year.
In short, UiPath turned in a robust quarter, with all key metrics moving solidly in the right direction. Investors had good reasons to send shares significantly higher on Friday.
Beth McKenna has no position in any of the stocks mentioned. The Motley Fool has positions in and recommends UiPath. The Motley Fool has a disclosure policy.
Coinbase Q3 revenue falls 5% to $674 million, but still beats expectations

Coinbase has released its shareholder letter for the third quarter of 2023, ending September 30th. Total revenue was $674 million, down 5% from last quarter. Net revenue was $623 million, down 6% sequentially. The company posted a small net loss of $2 million and generated positive Adjusted EBITDA of $181 million for the third consecutive quarter.
The crypto market continued to see low volatility and trading volumes in Q3. Bitcoin prices declined 12% from the end of last quarter, and crypto asset volatility hit multi-year lows. As a result, Coinbase’s transaction revenue fell 12% to $289 million. Consumer transaction revenue declined 11% to $275 million as trading volumes decreased 21%. Institutional transaction revenue dropped 18% to $14 million due to a 17% decline in trading volumes.
Subscription and services revenue was $334 million, largely flat from last quarter. Stablecoin revenue grew 14% to $172 million due to higher interest rates. However, blockchain rewards revenue fell 15% to $74 million driven by lower rewards rates and crypto prices.
Coinbase maintained a strong balance sheet ending Q3 with over $5.5 billion in cash, cash equivalents, USDC and custodial account overfunding. The company continued buying back debt at a discount, repurchasing $263 million of its 2031 Senior Notes for $177 million in cash during the quarter.
On the product side, Coinbase made advancements across its three pillar strategy. For crypto as an asset class, it added new assets, unified USD and USDC order books, and expanded internationally. For crypto updating the financial system, Coinbase entered an updated arrangement with Circle around USDC. And for crypto powering the Internet, it launched its Layer 2 scaling solution Base which saw over 10 million NFTs minted during its launch.
Regulatory clarity remains a top priority. Coinbase believes most G20 nations are adopting crypto regulations. It sees the EU’s MiCA regulation as a model framework and picked Ireland as its MiCA hub. In the US, the SEC case is proceeding on schedule with oral arguments set for early 2024.
For Q4, Coinbase expects subscription revenue to be flat sequentially. It anticipates lower stock-based compensation will drive expenses lower by $100-150 million versus Q3. The company believes it will generate meaningful positive Adjusted EBITDA for the full year 2023, an improvement from its prior goal. Despite ongoing volatility, Coinbase remains confident in the long-term potential of crypto.
The recent work stoppage by the United Auto Workers (UAW) union impacted the company’s ability to meet its financial targets.
On Thursday, October 26, Ford Motor Co (NYSE: F) reported its Q3 2023 earnings while missing Wall Street expectations. The company has been restructuring its operations and regrouping after the conclusion of nearly six weeks of US labor strike that cost the company a staggering $1.3 billion.
Ford in Q3 2023
In the third quarter, Ford reported a shift to net income, posting $1.2 billion, equivalent to 30 cents per share, compared to a loss of $827 million, or 21 cents per share, in the previous year. After accounting for specific items, the per-share earnings reached 39 cents.
The company experienced an 11% rise in overall revenue for the quarter, reaching $43.8 billion, up from $39.39 billion in the same period the prior year. Furthermore, adjusted earnings before interest and taxes (EBIT) showed a 22% increase, reaching $2.2 billion, compared to the previous year’s figure.
Soon after the results, the Ford stock tanked by more than 4% in the after-market hours. Ford has retracted its previously announced earnings forecast due to the recent work stoppage by the United Auto Workers (UAW) union, which ended with a tentative agreement.
The strike, which commenced on September 15, impacted the company’s ability to meet its financial targets. Before the strike, Ford was on track to achieve its earnings guidance, according to Chief Financial Officer John Lawler.
However, third-quarter results fell short of expectations, with adjusted earnings per share at 39 cents compared to the anticipated 45 cents, and automotive revenue at $41.18 billion against the expected $41.22 billion. Lawler attributed the underperformance to the UAW strike and ongoing cost and quality challenges that have affected the company’s operations in recent years. During the results call, Lawler told reporters:
“It is the cost and quality that we need to continue working on to improve the business. There’s a lot of positives within the business, and, unfortunately, it’s really not all shining through because of our cost and quality.”
Ford’s to Delay Its EV Plans
During the quarter, Ford’s traditional business operations under Ford Blue generated earnings of $1.72 billion, while its Ford Pro commercial business brought in $1.65 billion in revenue. On the other hand, the Model E electric vehicle unit reported a loss of $1.33 billion in the period from July to September.
Ford has decided to postpone approximately $12 billion in previously announced electric vehicle (EV) investments, including the construction of an EV battery plant in Kentucky. However, the company remains committed to progressing with its new EV plant and campus in West Tennessee, known as Blue Oval City.
The decision to delay these investments is due to lower-than-expected demand for EVs, coupled with rising raw material and labor costs, as well as pricing competition from leading EV manufacturer Tesla Inc (NASDAQ: TSLA).
“The transition to EVs is well underway. Adoption is growing, even if pace is slower than what the industry, including us, expected. Along the way, we’re going to balance production of gas, hybrid and electric vehicles in ways that many companies can’t, based on what consumers want,” said Lawler.
next
Business News, Market News, News, Stocks, Technology News

Bhushan is a FinTech enthusiast and holds a good flair in understanding financial markets. His interest in economics and finance draw his attention towards the new emerging Blockchain Technology and Cryptocurrency markets. He is continuously in a learning process and keeps himself motivated by sharing his acquired knowledge. In free time he reads thriller fictions novels and sometimes explore his culinary skills.
You have successfully joined our subscriber list.
Philips Raises Full-Year 2023 Expectations after Impressive Q3 Results
In light of its stellar performance and the resilience displayed in the face of challenges, Philips looks to uphold its commitment to creating enduring value and delivering sustainable impact in the long term.
Philips, a prominent Dutch health technology company, has exceeded market expectations in its 2023 third-quarter performance, leading to an upward revision of its full-year outlook.
The company’s core profit more than doubled, reaching an impressive 457 million euros ($483.3 million), while its comparable sales observed a significant 11% surge, totaling 4.5 billion euros.
Philips Raises FY Outlook for 2023
In an official press release on October 23, Philips attributed the solid performance to increased demand for medical scanners, patient monitoring equipment, and personal health services.
The company has adjusted its financial projections for the full year in response to its improved performance and solid order book despite acknowledging the uncertainties stemming from the volatile geopolitical environment.
“Based on Philips’ improved performance year-to-date, the strong order book, and the ongoing actions, the company is further raising its outlook for the full year 2023, although recognizing uncertainties remain in an increasingly volatile geopolitical environment,” it said.
Philips is now anticipating a growth of 6-7% in comparable sales and an adjusted EBITA profit margin of 10-11% for the year 2023. The Dutch technology company aims to achieve free cash flow at the upper end of the targeted range, between EUR 0.7-0.9 billion.
The revised FY guidance reflects an upward revision from its previous forecast of mid-single-digit sales growth and a high single-digit profit margin.
Philips to Diversify Chip Suppliers
While the demand for its medical products fueled sales, new orders experienced a decline of 9% compared to the previous year, primarily due to subdued demand from China post the pre-pandemic surge, compounded by persistent supply chain challenges.
To counter these challenges, the company’s CEO Roy Jakobs revealed a strategic shift towards local production in China and the diversification of chip suppliers, aiming to mitigate the impact of escalating trade tensions.
The company’s robust third-quarter performance, which exceeded the forecasts of analysts polled by the company, has been instrumental in fortifying its position and instilling confidence in its ability to navigate the dynamic market landscape.
With a strengthened order book and a focused approach toward improving operational efficiency, the company remains poised for sustained growth despite persistent uncertainties in the global geopolitical arena.
“We have witnessed a remarkable improvement in our operational performance, driven by our unwavering commitment to enhancing patient safety, reinforcing the reliability of our supply chain, and establishing a streamlined operational model,” said Jakobs.
He further noted that the company’s sustained emphasis on innovations in predictive data analytics and artificial intelligence across its diverse portfolio has yielded a positive impact, fostering an environment conducive to enhancing the quality and efficiency of care delivery.
In light of its stellar performance and the resilience displayed in the face of challenges, Philips looks to uphold its commitment to creating enduring value and delivering sustainable impact in the long term.
next
Business News, Market News, News, Stocks

Chimamanda is a crypto enthusiast and experienced writer focusing on the dynamic world of cryptocurrencies. She joined the industry in 2019 and has since developed an interest in the emerging economy. She combines her passion for blockchain technology with her love for travel and food, bringing a fresh and engaging perspective to her work.
You have successfully joined our subscriber list.